Nora Pecha, PharmD; Mathew A. Letizia, PharmD; Abir T. El-Alfy, PhD, MS
WMJ. 2026;125:P1. Published online March 31, 2026.
On January 30, 2025, the US Food and Drug Administration (FDA) approved Journavx (suzetrigine), a non-opioid analgesic, for the management of moderate-severe acute pain. Journavx is the first drug to be approved that targets the voltage-gated sodium channel NaV1.8. This novel target marks a new therapeutic class of non-opioid analgesics.
JOURNAVX MECHANISM OF ACTION
Journavx is a selective blocker of the voltage-gated sodium channel NaV1.8. This channel is selectively expressed in the peripheral pain-sensing neurons, including dorsal root ganglia, playing a role in pain signal transmission in the peripheral nervous system. The drug binds to the channel’s second voltage sensing domain and stabilizes the closed state. This results in tonic inhibition of the channel and reduces pain signal transmission. Lack of NaV1.8 expression in the central nervous system offers a favorable safety profile, without the addictive potential associated with opioid analgesics.
FDA-APPROVED INDICATION
- Treatment of moderate to severe acute pain in adults.
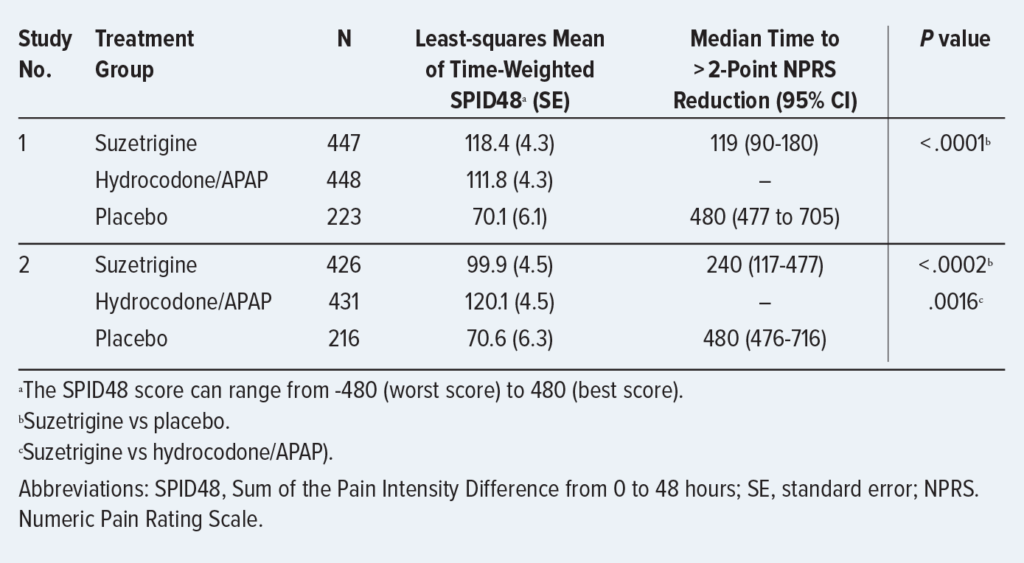
CLINICAL TRIAL DATA
FDA approval was based on the results of two randomized trials that evaluated Journavx efficacy. The trials are summarized below. Based on current safety data, Journavx is only approved for a maximum treatment duration of 14 days.
DOSING AND ADMINISTRATION
Recommended Dosing
- Loading dose: 100 mg once
- Maintenance dose: 50 mg every 12 hours
Administration
- Do not crush or chew
- Loading dose: take on an empty stomach, 1 hour before or 2 hours after food
- Maintenance dose: take without regard to food
Cost
- Average wholesale price: $15.50 per tablet
- Cost: $217
ADVERSE EFFECTS
The following adverse reactions have been reported:
- Skin rash (1%)
- Increased creatine phosphokinase in blood specimen (1% to 3%)
- Muscle spasm (1%)
- Decreased estimated glomerular filtration rate (eGFR) (3%)
CONTRAINDICATIONS
- Concomitant use of strong CYP3A4 inhibitors
DRUG/FOOD INTERACTIONS
Journavx is primarily metabolized by CYP3A4 enzyme. The metabolites are renally excreted (44%). Approximately 9.1% unchanged drug as well as the remaining metabolites are excreted in feces.
- Use of CYP3A4 inhibitors may increase the serum concentration of Journavx. Examples of strong CYP3A4 inhibitors include grapefruit juice, ketoconazole, clarithromycin, cyclosporine, and ritonavir.
- Use of CYP3A4 inducers may reduce the serum concentration of Journavx. Examples of strong CYP3A4 inducers include carbamazepine, phenytoin, rifampin, and primidone.
- Journavx may interact with some hormonal contraceptives. Patients using progestins, levonorgestrel, and norethindrone contraceptives should use additional nonhormonal contraception or alternative contraceptives during Journavx treatment and for 28 days after the last dose.
- Administration of the 100 mg loading dose with food decreases initial serum concentration. Subsequent maintenance doses are not affected by food.
CONCLUSIONS
The approval of Journavx marks a significant milestone in the development of non-opioid analgesics. Targeting the unique voltage-gated sodium channel NaV1.8 offers a novel therapeutic option for pain treatment and addresses the high need for non-opioid analgesics. Studies show that Journavx is effective in alleviating moderate to severe acute pain with no evidence of dependence or addiction potential. Further studies are warranted to examine its effect in chronic pain management.